Phase Change - Boiling Water Lab
Boiling water explanation:
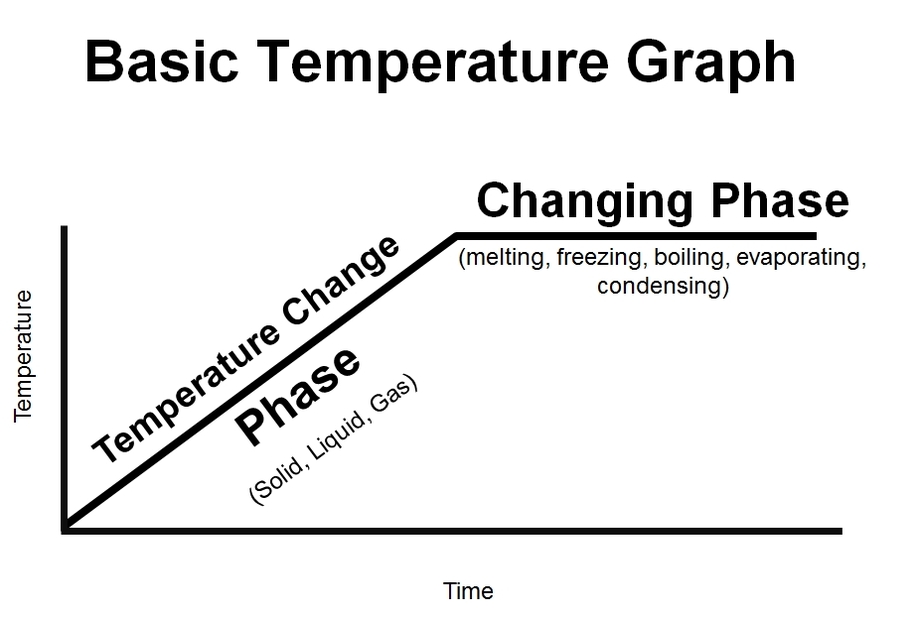
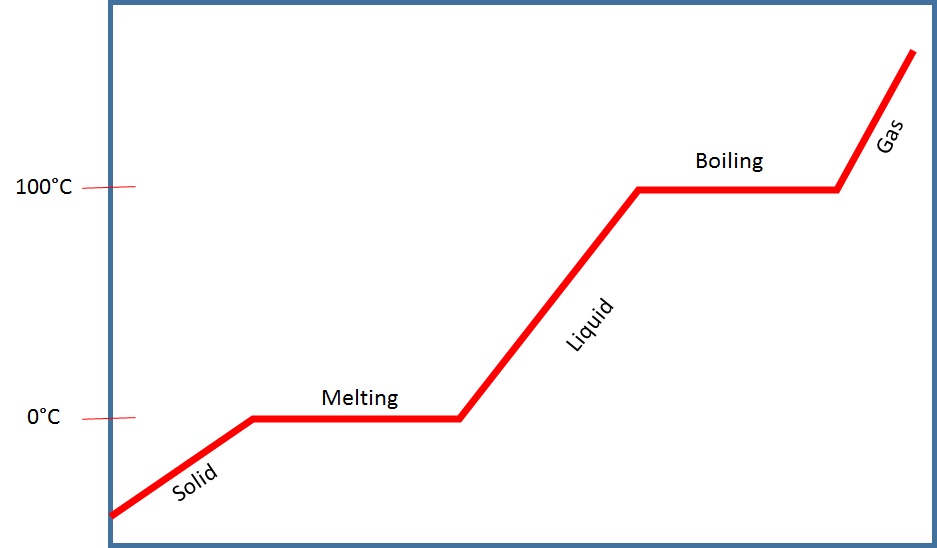
What you are seeing is the temperature change and what a phase and a phase change looks like on a temperature v. time graph.
Just like a distance v. time graph, a diagonal line means that the temperature is changing over time. However, a flat line, means that the temperature is not changing - but rather, the phases are changing.
The reason for this...
Let's take water as our example.
When water is below 0°C, the water is solid. What this means is that the bonding attraction between each of the water molecules are stronger than the vibration of the molecules due to the low level of heat energy.
As you put heat into the system, once it reaches 0°C the temperature stops even though there is heat energy going into the system.
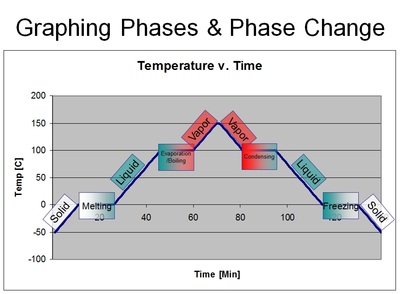
As it experiences a change in phase, such as melting, evaporating, boiling, condensation & freezing, the substance undergoes a phase change (hence a change in phase).
What you are seeing is the temperature change and what a phase and a phase change looks like on a temperature v. time graph.
Just like a distance v. time graph, a diagonal line means that the temperature is changing over time. However, a flat line, means that the temperature is not changing - but rather, the phases are changing.
The reason for this...
Let's take water as our example.
When water is below 0°C, the water is solid. What this means is that the bonding attraction between each of the water molecules are stronger than the vibration of the molecules due to the low level of heat energy.
As you put heat into the system, once it reaches 0°C the temperature stops even though there is heat energy going into the system.
As it experiences a change in phase, such as melting, evaporating, boiling, condensation & freezing, the substance undergoes a phase change (hence a change in phase).
To explain what you are seeing - and why you are seeing it is explained in the following:
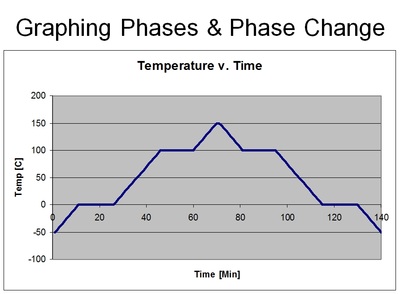
Anytime you see a diagonal line - it means change. However, in this case, a change in what? In this case, a change is a change in temperature. So be careful when you see a flat line - because it too means change - but rather a change in phases! In other words, the temperature of the substance is stuck until the phase of the substance has completely changed into another phase. In other words, the liquid water was stuck at 100 degrees Celsius until every last drop of water was converted to steam. Once it is all converted, the temperature can continue to rise.
The reason for this is explained by the pictures below.
Anytime you see a diagonal line - it means change. However, in this case, a change in what? In this case, a change is a change in temperature. So be careful when you see a flat line - because it too means change - but rather a change in phases! In other words, the temperature of the substance is stuck until the phase of the substance has completely changed into another phase. In other words, the liquid water was stuck at 100 degrees Celsius until every last drop of water was converted to steam. Once it is all converted, the temperature can continue to rise.
The reason for this is explained by the pictures below.
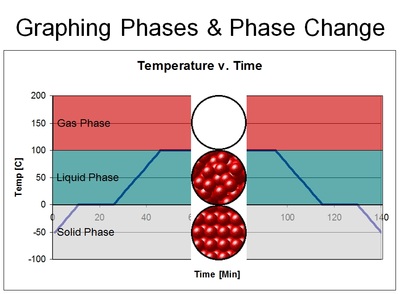
Each phase of matter is dependent upon the amount of thermal/heat energy the substance has in relation to the strength of the bonds between molecules found within the substance.
Solids, for instance, has a relatively low amount of thermal energy when compared to the strength of the bonds between each set of molecules found within the substance. In other words, the attraction between the water molecules in solid ice is stronger than the vibration caused by heat. Because the bonds are not broken, solids tend to have a definite shape and volume.
Liquids, however, has a relatively higher level of thermal energy when compared to the strength of the bonds between water molecules. In fact, there is enough energy to break the bonds between the water molecules, but the attraction between the water molecules is strong enough to reform. So in liquids, bonds are forming and breaking all the time. This is what creates that fluid dynamic - where liquids have no definite shape, but it does have a definite volume.
Gasses, on the other hand, the bonds have been entirely broken due to the level of energy being great enough to break them.
Phases v. Phase Change.
The reason why the temperature stops - even though in our boiling water lab experiment the fire was completely on the whole time - is because the heat energy that is being pumped into the water is going into breaking the bonds between the atoms instead of making the atoms vibrate faster. And that's why you'll see a "temperature change" (such as solid, liquid, gas) as a diagonal line, and a "phase change" (such as melting/freezing, boiling, evaporation & condensation) as a flat line. See the following graphs below.
Solids, for instance, has a relatively low amount of thermal energy when compared to the strength of the bonds between each set of molecules found within the substance. In other words, the attraction between the water molecules in solid ice is stronger than the vibration caused by heat. Because the bonds are not broken, solids tend to have a definite shape and volume.
Liquids, however, has a relatively higher level of thermal energy when compared to the strength of the bonds between water molecules. In fact, there is enough energy to break the bonds between the water molecules, but the attraction between the water molecules is strong enough to reform. So in liquids, bonds are forming and breaking all the time. This is what creates that fluid dynamic - where liquids have no definite shape, but it does have a definite volume.
Gasses, on the other hand, the bonds have been entirely broken due to the level of energy being great enough to break them.
Phases v. Phase Change.
The reason why the temperature stops - even though in our boiling water lab experiment the fire was completely on the whole time - is because the heat energy that is being pumped into the water is going into breaking the bonds between the atoms instead of making the atoms vibrate faster. And that's why you'll see a "temperature change" (such as solid, liquid, gas) as a diagonal line, and a "phase change" (such as melting/freezing, boiling, evaporation & condensation) as a flat line. See the following graphs below.