Mini Science Fair introduction
Each 8th Grade student will be required to conduct an individual experiment at home during the first semester this year. Students will be required to select an experiment from sciencebuddies.org., conduct the experiment, and hand in a comprehensive write-up for that experiment.
Students will be graded both periodically based on completion of individual sections of the experiment and comprehensively upon completion of the experiment and the final project write-up.
The student will be provided with instructions in class throughout the semester on how to complete each individual section and with a cover sheet detailing each required element for that section to complete at home.
We ask that the completed assignments be typed. If your student does not have access to a computer at home, please speak with your teacher about alternate accommodations.
Students who wish to continue on with their experiment and possibly participate in the School/District Science Fair should talk with their teacher about the requirements for this extension.
Students will be graded both periodically based on completion of individual sections of the experiment and comprehensively upon completion of the experiment and the final project write-up.
The student will be provided with instructions in class throughout the semester on how to complete each individual section and with a cover sheet detailing each required element for that section to complete at home.
We ask that the completed assignments be typed. If your student does not have access to a computer at home, please speak with your teacher about alternate accommodations.
Students who wish to continue on with their experiment and possibly participate in the School/District Science Fair should talk with their teacher about the requirements for this extension.
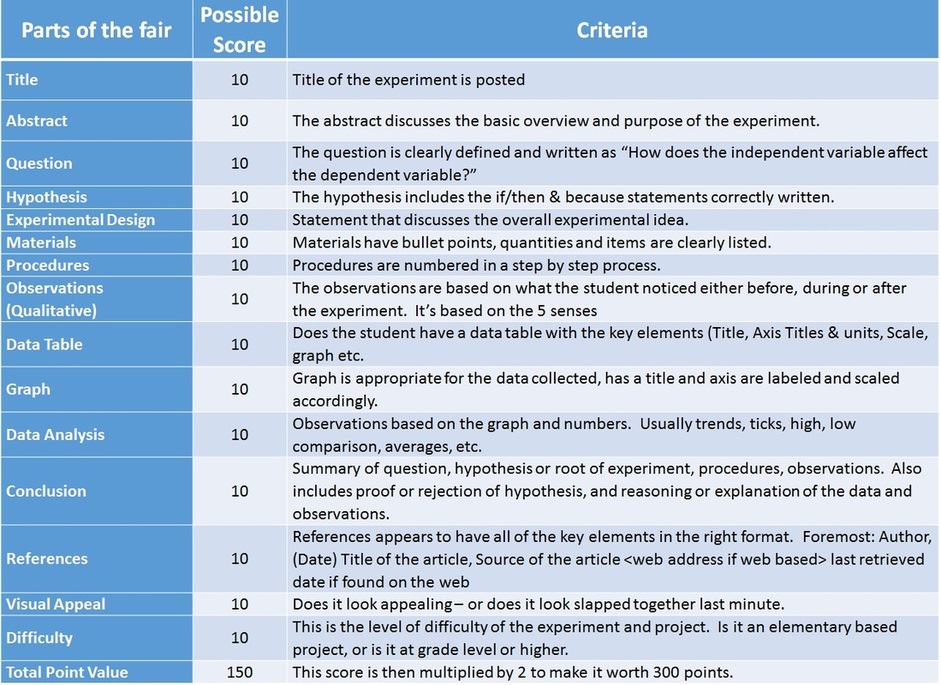
Science Fair Cover Sheets
Link to the cover sheets
Science Fair Poster Example
This is an example of what your poster could look like. It's simple, straight forward, and includes the key elements of each aspect of your science fair project that is laid out in a book like fashion. Below is the scoring rubric that will be used during the evaluation of your science fair project.
Rubric Score Guide
* After scoring each element - the final score is multiplied by 2 to make it worth 300 points.
Starting Your Science Fair Project
If you don't have a question to research and wonder what you should do for your science fair project, consider first what are your hobbies or areas of interest. Next consider a more specific area of that area of interest - and what is something you can change about it.
If you're still struggling - consider going to www.sciencebuddies.org - it will help you determine a good science fair project. Just make sure that it is an experiment - and not a report.
If you're still struggling - consider going to www.sciencebuddies.org - it will help you determine a good science fair project. Just make sure that it is an experiment - and not a report.
Science Fair Rules & Parameters
- It must be core specific. This means that your science fair must be related to the following fields of scientific study: Physics, Chemistry, Geology & Ecology.
- It must be 8th grade level based questions or higher.
- It cannot be similar to a lab we've already done in class.
- There will be no Mento's and Coke experiments.
- There will be no experiments on animals or humans.
- There must be a scientific explanation behind every experiment and cited as such.
|
Physics Concepts
|
Chemistry Concepts
|
Geology Concepts
|
Ecology Concepts
|
Science Fair Basic Lab Write Up Example with Key Elements
|
Abstract
In this experiment we will examine the overall properties of water - in particular, cohesion and adhesion. To do this we will investigate how many drops of pure water a penny can hold and compare it to how many drops of water the penny will hold if we inject different materials into the water - such as soap, vinegar, dirt, salt and food dye.
Since cohesion is what allows water to form a dome (Perlman, 2014) - we wanted to see if the different materials affect the cohesion of water. We think that it does and that it will diminish the number of drops of water that a penny can hold due to the dissolved particulates or molecules slipping in between the water molecules - which disrupts the attraction between water molecules. Experimental Question and Hypothesis: Question: How does salt affect the number of drops of water a penny can hold Hypothesis: If I add 10g of salt to 200 mL of water, then the number of drops a penny can hold will go down because the salt molecules will slip in between the water molecules - which decreases the attraction between each other. Experimental Design: To test this, dissolve 10g of salt into 200mL of water and test to see how many drops of salt water a penny can hold and compare it to the amount of drops of regular water a penny can hold. Materials:
Procedures:
Observations:
|
Abstract
Experimental Question & Hypothesis:
Experimental Design:
Materials
Procedures
Observations:
|
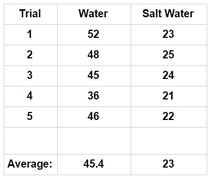
Data:
|
*(Please note that this data is made up and may not reflect true results)
|
Data Table:
|
|
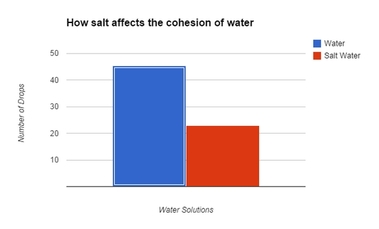
Graph:
The elements of the graph include the following:
|
|
Data Analysis:
Conclusion: In our research, we were investigating how salt affects the cohesive properties of water. To test this, we were using the number of drops a penny can hold as our indicator for cohesiveness after we added salt to the solution. According to our data, by adding salt to the water seemed to affect the cohesive properties of the water, as indicated by the severe reduction of the number of drops the penny would hold. We were not too surprised with our findings. We did notice some anomalies during our experiment, however. For instance, we did notice that we have a wide range of drops with the pure water solution, which ranged from 36-52. We think the lower number is more of the exception than the rule - as we noticed that the next number in series was closer to the higher number than the lower number. We think it might be because as we dropped the water, the drops may have been slightly bigger, or the drops touched the puddle on the penny, which reduced the number of drops. If we were to run this test again, we need to increase the height the drops that were administered to ensure that the drops don’t come in contact with the puddle while the water is still being formed from the pipet. The salt water solution, however, has a much tighter range of data - only having a four point difference and each of them in the lower to mid 20’s. What this means is that our hypothesis is thus far correct - in that the salt does seem to disrupt the cohesive properties of water. We still think that the reason the salt disrupts the cohesive property of water is due largely in part to the salt molecules slipping in between and separating the water molecules. We do know that water is polar and the salt becomes ionic when dissolved in water (Ophardt, 2003). Perhaps the adhesive attraction that water has with the salt is stronger than the cohesive property of water (Freeman, 2007). Further testing would be needed to suggest such a hypothesis; therefore, to further examine to see if cohesion is disrupted with additives dissolved in water, I would suggest that we test to see how sugar and soap affects waters ability to cohesively stick to it's self and to adhesively stick to other materials. |
Data Analysis:
Conclusion: This is the biggest, most important part of the experiment. This is where you synthesize the whole project into a short essay - this portion touches on each portion of the write up by addressing the following bullet points:
|
|
References
Perlman, H. (March 17, 2014) “Adhesion & Cohesion of Water” USGS.gov <http://water.usgs.gov/edu/adhsion.html> Last Retrieved August 18, 2014
Ophardt, C. (2003) “Solubility of Salts” Virtual Chembook, Elmhurst College <http://www.elmhurst.edu/~chm/vchembook/171solublesalts.html> Last Retrieved September 9, 2014 Freeman, S. (October 18, 2007) “How Water Works” HowStuffWorks.com <http://science.howstuffworks.com/environmental/earth/geophysics/h2o7.htm> Last Retrieved August 18, 2014 |
References
Author Last, F. (Date Published) "Title of the article written" Publisher Source <full web address> Last Retrieved Date
|