Chemical & Physical Properties
Chemical & Physical Properties (PPT)
Chemistry is the study of chemical & physical properties and changes of matter.
Matter is anything that has mass (stuff that is measured in grams) and takes up space (volume that is measured in Liters). In other words, it's all the atoms that make up the object.
Substance: a single type of matter that is pure, has a specific make up or composition.
i.e. Oxygen (O2), Water (H2O), Salt (NaCl)
Mixture: more than one substance together.
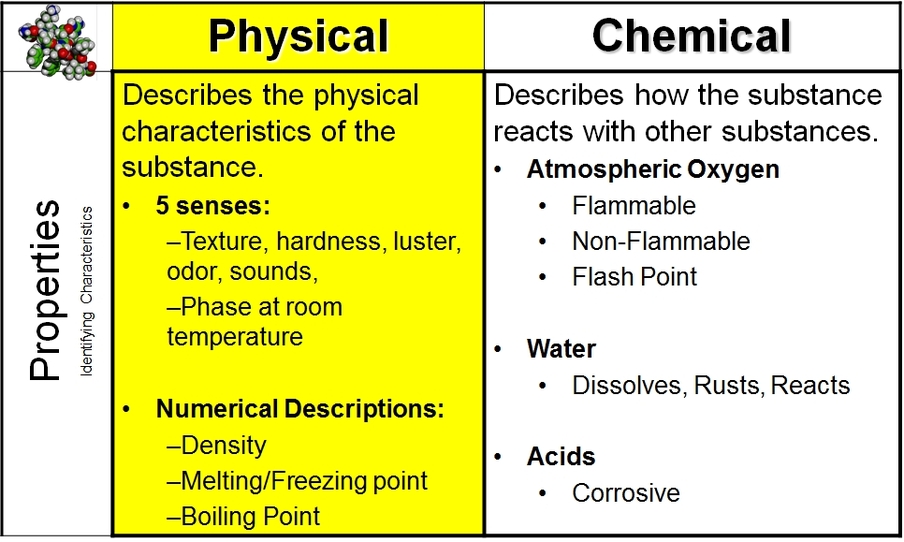
To identify any substance you use properties. There are two different types of properties; chemical and physical.
•Physical Property: Describes the physical characteristics / attributes of the substance.
•Qualitative: what the substance looks like, feels like, tastes like, smells like, sounds like.
•Quantitative: numerical properties such as phase at certain temperatures, density, melting/freezing point, etc.
Describing the physical property of a substance is like describing a person. He's got red hair, green eyes, 5' 6" etc. i.e. flour is white & powdery; sugar is white and granular.
•Chemical Property: How a substance reacts with other substances.
i.e. Iron reacts with Oxygen to produce Rust.
Silver reacts with Sulfur to produce tarnish - a gray looking substance.
Baking soda reacts with vinegar to produce carbon dioxide
Sugar, when heated, will caramelize
Sometimes two substances may look an awful lot a like. This is like two twins who dress up a like - but have different personalities. One way to tell which twin is which is by how they interact with other people to determine which twin it is.
With chemicals that look a lot alike, like sugar, salt and citric acid for instance, w/o tasting it, you will want to rely on chemical properties - such as how does it behave or react with other substances or heat.
To identify which one is sugar based on physical properties alone can be a bit difficult. However, you can tell them apart by how they interact with other substances. One test that you could do is see what they do when heated - as we know that sugar caramelizes w/ heat; citric acid and salt do not.
Another way to describe chemical properties is to react it with another substance. I.e. citric acid, when diluted with water, will react with baking soda to produce carbon dioxide and some other substance.
Chemistry is the study of chemical & physical properties and changes of matter.
Matter is anything that has mass (stuff that is measured in grams) and takes up space (volume that is measured in Liters). In other words, it's all the atoms that make up the object.
Substance: a single type of matter that is pure, has a specific make up or composition.
i.e. Oxygen (O2), Water (H2O), Salt (NaCl)
Mixture: more than one substance together.
To identify any substance you use properties. There are two different types of properties; chemical and physical.
•Physical Property: Describes the physical characteristics / attributes of the substance.
•Qualitative: what the substance looks like, feels like, tastes like, smells like, sounds like.
•Quantitative: numerical properties such as phase at certain temperatures, density, melting/freezing point, etc.
Describing the physical property of a substance is like describing a person. He's got red hair, green eyes, 5' 6" etc. i.e. flour is white & powdery; sugar is white and granular.
•Chemical Property: How a substance reacts with other substances.
i.e. Iron reacts with Oxygen to produce Rust.
Silver reacts with Sulfur to produce tarnish - a gray looking substance.
Baking soda reacts with vinegar to produce carbon dioxide
Sugar, when heated, will caramelize
Sometimes two substances may look an awful lot a like. This is like two twins who dress up a like - but have different personalities. One way to tell which twin is which is by how they interact with other people to determine which twin it is.
With chemicals that look a lot alike, like sugar, salt and citric acid for instance, w/o tasting it, you will want to rely on chemical properties - such as how does it behave or react with other substances or heat.
To identify which one is sugar based on physical properties alone can be a bit difficult. However, you can tell them apart by how they interact with other substances. One test that you could do is see what they do when heated - as we know that sugar caramelizes w/ heat; citric acid and salt do not.
Another way to describe chemical properties is to react it with another substance. I.e. citric acid, when diluted with water, will react with baking soda to produce carbon dioxide and some other substance.