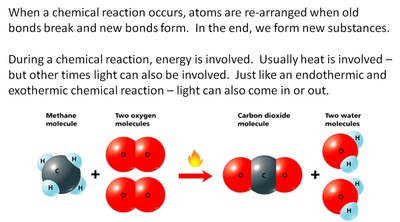
Photosynthesis & Respiration
- What patterns do you see concerning the sunflower?
- What allows it to move and why does it follow the sun?
- What purpose does it serve?
Sunflowers, like all plants, need the sun to live. (But so do you! And you're not even a plant! But that's for another story.) The plant, unlike you, needs the sun to make it's own food! Wha????
What is photosynthesis anyway? Do we really need it to live?
Yes, sunflowers make their own food using the light of the sun. And without it, you wouldn't be able to live! That's right! Without those little green plants, you're life would cease to exist. Plants undergo a chemical reaction called photosynthesis to make a couple of common chemicals that you and I depend on - that's right... Glucose, Fructose and Sucrose - also known as... "Sugar!"
Ironically, it also makes another chemical that you and I depend on... Oxygen! Yes, we use both of those chemicals that plants make in order for you and I to live. We breath in the oxygen and eat the sugars that plants make.
These two chemicals pack a punch too! Yep. They contain energy. This energy, allows us to move around. It's like our own little battery. They make it, we take it.
This is how it all works:
Ironically, it also makes another chemical that you and I depend on... Oxygen! Yes, we use both of those chemicals that plants make in order for you and I to live. We breath in the oxygen and eat the sugars that plants make.
These two chemicals pack a punch too! Yep. They contain energy. This energy, allows us to move around. It's like our own little battery. They make it, we take it.
This is how it all works:
- Plants bring up water up the roots and absorb carbon dioxide into their leaves.
- These chemicals meet up in a plants cell where the chlorophyll is and whamo! It's zapped from the light from the sun and whala, out comes sugar and oxygen!
- Oxygen and Sugar are full of energy that the plant used from the sunlight.
- We eat the sugar and breath in the oxygen - where these two chemicals meet up in our own cells at a site called the mitochondria.
- The mitochondria take apart the two chemicals and in the process, removes the energy so that your body can function, and makes carbon-dioxide and water. This process is called RESPIRATION. Yes, Respiration is a chemical reaction. No, it is not all about your lungs or about breathing. That's part of the Respiratory System that removes the carbon dioxide and water from your body! So don't get it confused - okay!
- Carbon Dioxide and Water have very little chemical energy. But these chemicals are returned to the plants so that it can be re-zapped back into higher level energy chemicals with light and turn back into Oxygen and Sugar. This is where the whole process starts all over again.
Photosynthesis / Respiration Class Activity
Instructions:
- Split the class into groups of at least 5.
- Hand each group a Reactants Card, a Products Card, 36 Toothpicks, 18 Gumdrops, 6 Large Marshmallows, 12 Small Marshmallows, a timer, and a Flashlight.
- Explain the Key on the bottom of each card and model how to use the toothpick to create bonds. Show an example of how to make each molecule using the guide on the cards.
- Explain that they will be having a contest with the groups to see how many times they can cycle through the photosynthesis/respiration cycle in 10 minutes. Have one person in the group record how many times they can finish the cycle in the given time.
- Split the groups in half and have half of a group work as the photosynthesis side of the cycle and half work as the respiration side. Have the halves move to opposite lab stations (or tables if needed) across the room from each other.
- The photosynthesis group will begin the process. They will have all the supplies at their station. They will construct/produce 1 glucose molecule and 6 oxygen molecules using the photosynthesis-products card as a guide. One member of the group will have to shine the flashlight on the group to simulate the sun and the energy needed to initiate photosynthesis. Have this same person record the number of complete cycles and set the timer for 10 minutes. Start the timer. After the group has finished constructing the 1 glucose molecule and 6 oxygen molecules, they will then signal to the respiration group who will send one from their group to come and retrieve the finished product. (This simulates the animal having to move in order to eat the plant since the plant cannot move itself.) The respiration group will then the take the products over to their lab station.
- The respiration group will now deconstruct the glucose and oxygen molecules. Explain that this process represents the breaking of chemical bonds and the release of chemical energy which is then converted by the organism into mechanical and heat energy. After breaking apart the molecules, they will then proceed to construct 6 Carbon Dioxide Molecules and 6 Water Molecules using the Respiration-Products Card as a guide. They will then run their completed respiration products back over to the photosynthesis group who will take apart the respiration products. (Again, simulating how the plants cannot move.)
- The students will repeat steps 4-5 until the time is up. Have each group tally how many times they completed the entire process and compare their scores.
- Extension: Discuss possible problems that arose and have the students relate these problems to real-life situations. (For example: One group might complain about how hard it was to take apart the glucose molecules and they could relate it to how some foods are “harder” to break down than others and require more energy on the part of the organism, etc. Make sure and discuss the mobility issues surrounding plants vs. animals)
- Depending on the time, have the members of the groups switch positions and see if that will affect their tallies. Relate this to different species of plants and animals and the different efficiencies they have in producing and cycling energy.
Some questions to consider during this activity:
Photosynthesis is the process that uses ____________ energy from the sun, together with __________________ and ____________ to make ____________________ and oxygen.
Respiration is a process that converts ______________ energy in food (glucose) into ______________ energy and __________ energy. It produces ______________and __________.
The formula for Photosynthesis is:
Light + 6CO2 + 6H2O → C6H12O6 + 6O2
The formula for Respiration is:
C6H12O6 + 6O2 → 6CO2 + 6H2O
In this lab, you will be simulating the processes of photosynthesis and respiration. You will be constructing molecules from reactants to form the products of photosynthesis and respiration. Answer the following analysis questions after completing each section of the lab.
Part 1: Photosynthesis and Respiration
Photosynthesis is the process that uses ____________ energy from the sun, together with __________________ and ____________ to make ____________________ and oxygen.
Respiration is a process that converts ______________ energy in food (glucose) into ______________ energy and __________ energy. It produces ______________and __________.
The formula for Photosynthesis is:
Light + 6CO2 + 6H2O → C6H12O6 + 6O2
The formula for Respiration is:
C6H12O6 + 6O2 → 6CO2 + 6H2O
In this lab, you will be simulating the processes of photosynthesis and respiration. You will be constructing molecules from reactants to form the products of photosynthesis and respiration. Answer the following analysis questions after completing each section of the lab.
Part 1: Photosynthesis and Respiration
- Diagram the transfer of energy that occurs in photosynthesis.
- Diagram the transfer of energy in respiration.
- Trace the path of energy from sun to you.
- How many complete cycles was your group able to complete?______
- What were some problems you encountered as you replicated photosynthesis and respiration?
- How might these problems relate to the real-world? Are there any problems that might arise in the environment that could affect the construction/production of needed molecules?
- What was your environmental hazard?
- How did this affect your photosynthesis/respiration cycle?
- What were some of the other environmental hazards discussed?
- On your own, list other possible environmental hazards that an organism might encounter.
- How do you think these hazards will affect the cycle.
What happens if we remove some of the plants in an ecosystem?
Environmental concerns
Environmental Hazards Extension
- Continue to keep the classroom in their groups. Pass out an environmental hazard card to each group. Explain that each group has to follow the instructions on the card and permanently remove the molecules listed from their cycle when those items are produced. Have them try to complete the entire cycle at least one time without the items that were removed. Have them record in their packets how the hazard affected the photosynthesis/respiration process and answer the environmental hazards questions in their packet.
- Have the groups share their environmental hazard and explain to the class how it affected the cycling of energy and the photosynthesis/respiration process.
- Extension: Have the students think of other environmental hazards that could affect this cycle. Have them record their ideas in their packet and share with the class next time.
Some environmental hazards that occur in nature may include:
- EARLY FREEZE (unprepared animals may die off prematurely)
- NEW HERBIVORE SPECIES (Animal Eats Plants)
- URBAN DEVELOPMENT (Buildings Block Sunlight) Lose 1 Glucose (C6H12O6) Molecule
- DROUGHT (Lack of water affects both plants and animals)
- DISEASE (Affects the health and ability for a plant or animal to survive)
- EARLY WINTER (Many animals die from lack of preparation)
- OVERGRAZING (Cows eat all the grass before it has time to reproduce)
- INVASIVE SPECIES (New species takes over habitat of native plants)
- URBAN SPRAWL (Replaces forest with housing development)
Here's the more technical explanation of
photosynthesis & respiration
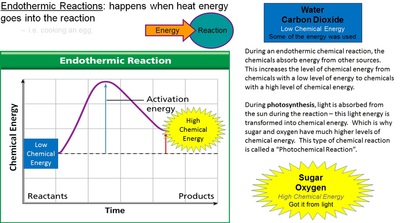
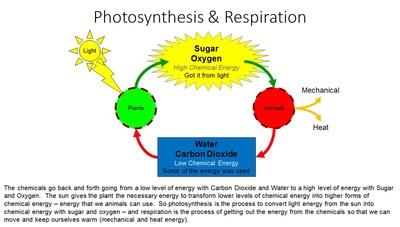
Photosynthesis reactions take chemicals that have low levels of chemical energy (water & carbon dioxide) and use the light energy from the sun to convert them into chemicals that have high levels of chemical energy (sugar & oxygen).
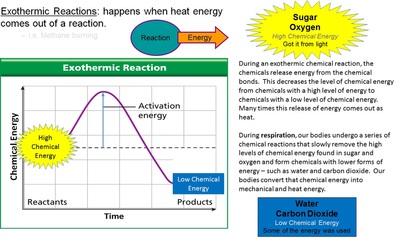
Respiration reactions take the chemicals that have high levels of chemical energy (sugar and oxygen) and extract the energy so that they can move (mechanical energy) and keep themselves warm (heat energy). In that process they make water and carbon dioxide. These chemicals go back to the plant to be re-energized.
These reactions are very similar to the mousetrap. A mousetrap that is set has a lot of pent up energy. But, once the trap is tripped, all of that pent up energy is released. In the end, you have a mouse trap that is at a lower form of pent up energy that will not snap any further. This is like the chemicals that are used between photosynthesis and respiration. We cycle between chemicals that have higher forms of energy and lower forms of energy and then use outside forms of energy (light) to put back that chemical energy - much like we use a hand to reset the trap.
Respiration reactions take the chemicals that have high levels of chemical energy (sugar and oxygen) and extract the energy so that they can move (mechanical energy) and keep themselves warm (heat energy). In that process they make water and carbon dioxide. These chemicals go back to the plant to be re-energized.
These reactions are very similar to the mousetrap. A mousetrap that is set has a lot of pent up energy. But, once the trap is tripped, all of that pent up energy is released. In the end, you have a mouse trap that is at a lower form of pent up energy that will not snap any further. This is like the chemicals that are used between photosynthesis and respiration. We cycle between chemicals that have higher forms of energy and lower forms of energy and then use outside forms of energy (light) to put back that chemical energy - much like we use a hand to reset the trap.
Photosynthesis: (Like Endothermic Rxn’s)
Plants take in Water and Carbon Dioxide. Plants take in Light energy from the sun and transform it into chemical energy by making sugar and Oxygen. Sugar and oxygen have a high level of chemical energy.
What qualifies any chemical as having more or less chemical energy is it's reactivity. If it gives off a lot of energy during reaction - that's because it has a lot of stored chemical energy.
You can test this idea out in a few ways.
This is like taking a mousetrap that is set and tripping it off.
Plants take in Water and Carbon Dioxide. Plants take in Light energy from the sun and transform it into chemical energy by making sugar and Oxygen. Sugar and oxygen have a high level of chemical energy.
What qualifies any chemical as having more or less chemical energy is it's reactivity. If it gives off a lot of energy during reaction - that's because it has a lot of stored chemical energy.
You can test this idea out in a few ways.
- What happens when you give a kid a lot of sugar? (Hint: they get hyper - because they have a lot of energy)
- What happens if you take powdered sugar and puff a cloud of it over a fire? (Hint: it creates a ball of fire)
- What does every fire need in order to react? (Hint: Heat, fuel & oxygen)
This is like taking a mousetrap that is set and tripping it off.
Respiration: (Like Exothermic Rxn’s)
Animals breath in Oxygen and eat the sugar from plants. Animals then metabolize the sugar and oxygen and convert it back into Carbon Dioxide and Water. This process releases the stored chemical energy and converts it into mechanical and heat energy.
Carbon Dioxide and Water do not have a lot of chemical energy. In fact, they are pretty stable. This is why it's considered lower levels of chemical energy - they're not very reactive. In fact, they are usually the end-product of most chemical reactions that stem from fire. In fact, water and carbon dioxide are usually the chemicals they use to put out fires.
Animals breath in Oxygen and eat the sugar from plants. Animals then metabolize the sugar and oxygen and convert it back into Carbon Dioxide and Water. This process releases the stored chemical energy and converts it into mechanical and heat energy.
Carbon Dioxide and Water do not have a lot of chemical energy. In fact, they are pretty stable. This is why it's considered lower levels of chemical energy - they're not very reactive. In fact, they are usually the end-product of most chemical reactions that stem from fire. In fact, water and carbon dioxide are usually the chemicals they use to put out fires.
Enrichment
This is a video about the chemistry and science behind food.
Here's something really cool about the mathematics behind sunflowers.