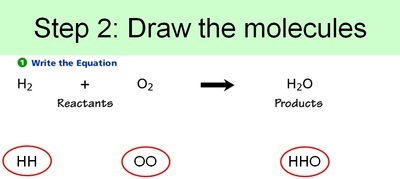
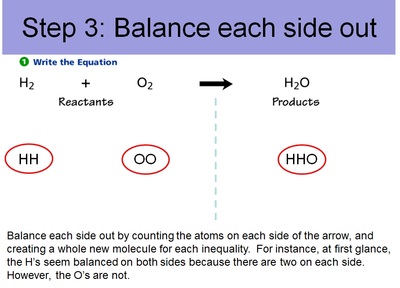
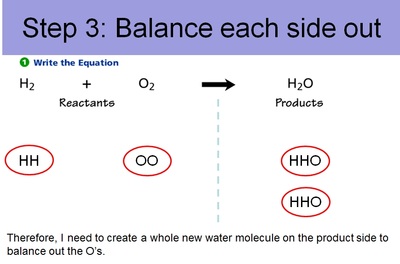
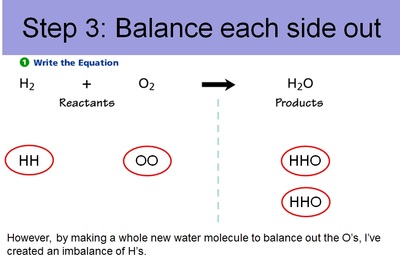
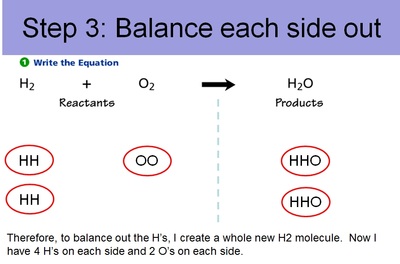
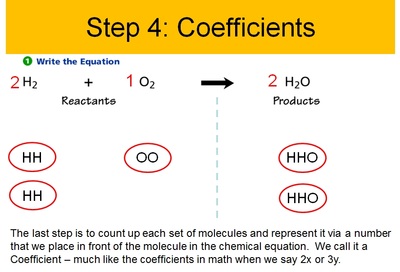
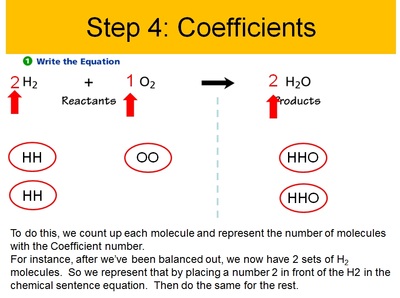
Conservation of Matter Allows for Balancing Chemical Equations
Balancing Equations:
The big idea for today:
The big idea for today:
- Matter or Mass is neither created nor destroyed.
- Because of that, we need to balance the chemical equation to find the right ratios in a chemical formula.
- Reactants --> Products
- Come see me on how to balance a chemical equation.
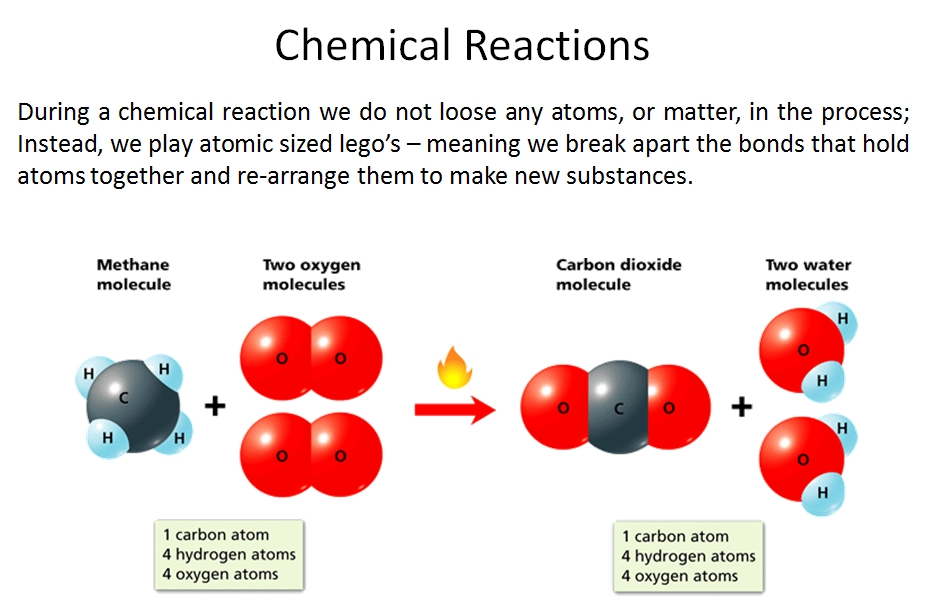
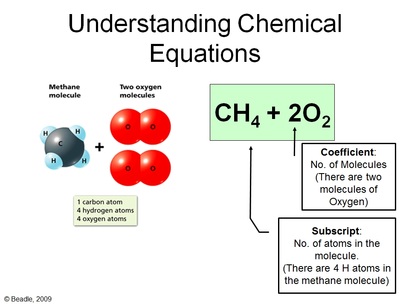
You'll notice here that we did not lose any atoms of carbon, oxygen or hydrogen in this chemical reaction. All we did was re-arrange them to form new substances. We reacted methane (CH4) with Oxygen (2 O2) and produced Carbon Dioxide (CO2) and Water (H2O). This principle is called Conservation of Matter; meaning, matter can neither be created nor destroyed - it's recycled!
Therefore, since you can't get rid of atoms or create new atoms, we need to account for every atom during a chemical reaction. And, just like making cookies, we need to know the right ratio's of ingredients. This entails balancing out chemical equations. Below are the steps to balancing out chemical equations.
Therefore, since you can't get rid of atoms or create new atoms, we need to account for every atom during a chemical reaction. And, just like making cookies, we need to know the right ratio's of ingredients. This entails balancing out chemical equations. Below are the steps to balancing out chemical equations.
Notes: CHEMICAL EQUATIONS
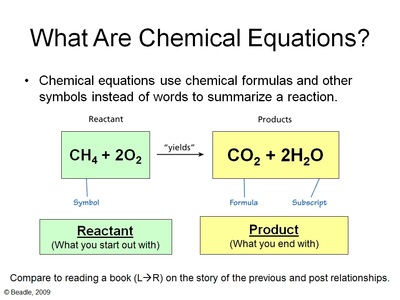
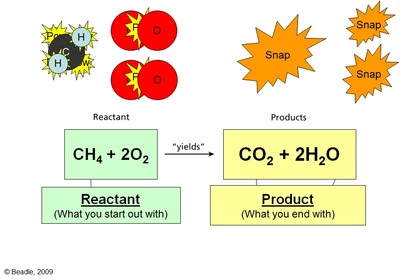
In every chemical reaction, there are participants known as reactants, which, by chemically reacting to one another, result in the creation of a product or products. As stated earlier, a chemical reaction involves changes in the arrangement of atoms. The atoms in the reactants (or, if the reactant is a compound, the atoms in its molecules) are rearranged. The atomic or molecular structure of the product is different from that of either reactant.
Note, however, that the number of atoms does not change. Atoms themselves are neither created nor destroyed, and in a chemical reaction, they merely change partners, or lose partners altogether as they return to their elemental form. This is a critical principle in chemistry, one that proves that medieval alchemists' dream of turning lead into gold was based on a fallacy. Lead and gold are both elements, meaning that each has different atoms. To imagine a chemical reaction in which one becomes the other is like saying "one plus one equals one."
Read more: http://www.scienceclarified.com/everyday/Real-Life-Chemistry-Vol-2/Chemical-Reactions-Real-life-applications.html#b#ixzz26JjuxVwh
In every chemical reaction, there are participants known as reactants, which, by chemically reacting to one another, result in the creation of a product or products. As stated earlier, a chemical reaction involves changes in the arrangement of atoms. The atoms in the reactants (or, if the reactant is a compound, the atoms in its molecules) are rearranged. The atomic or molecular structure of the product is different from that of either reactant.
Note, however, that the number of atoms does not change. Atoms themselves are neither created nor destroyed, and in a chemical reaction, they merely change partners, or lose partners altogether as they return to their elemental form. This is a critical principle in chemistry, one that proves that medieval alchemists' dream of turning lead into gold was based on a fallacy. Lead and gold are both elements, meaning that each has different atoms. To imagine a chemical reaction in which one becomes the other is like saying "one plus one equals one."
Read more: http://www.scienceclarified.com/everyday/Real-Life-Chemistry-Vol-2/Chemical-Reactions-Real-life-applications.html#b#ixzz26JjuxVwh