Lab Write up (computers)
Lab Write Up ExampleAbstract
From our previous experiment, we examined how many drops a penny can hold - and in our studies, we noticed that the tails side held more drops than the heads side. We feel that the main reason was that one of our colleagues held the dropper either closer or further from the penny.
In this experiment, we examine how height (independent variable) affects the number of drops a penny can hold (dependent variable). My hypothesis is that by increasing the height of the dropper, the drops will go down based on the idea that as you increased height, you also simultaneously increase the drops speed due to gravitational acceleration via gravitational potential energy. The experimental results confirmed my hypothesis by showing the decrease of drops the penny can hold as we increased height. Experimental Question and Hypothesis: Question: How does salt affect the number of drops of water a penny can hold Hypothesis: If I add 10g of salt to 200 mL of water, then the number of drops a penny can hold will go down because as the salt slips in between the water molecules, this may disrupt the cohesive strengths of water. Also, due to the strong polar nature of salt molecules when dissolved, water may have a harder time sticking to each other, making adhesion possibly a stronger force than cohesion while in solution. Experimental Design: To test this, dissolve 10g of salt into 200mL of water and test to see how many drops of salt water a penny can hold and compare it to the amount of drops of regular water a penny can hold. Materials:
Procedures:
Observations
|
Lab Write up elementsAbstract:
Experimental Question & Hypothesis:
Experimental Design:
Observations:
|
|
Data Collection and further observations:
|
|
Data Analysis:
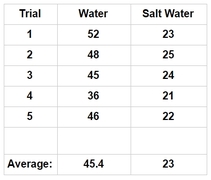
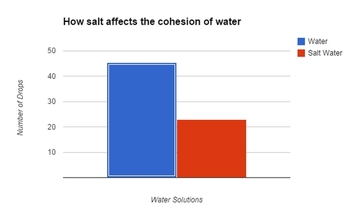
Conclusion: In our research, we were investigating how salt affects the cohesive properties of water. To test this, we were using the number of drops a penny can hold as our indicator for cohesiveness after we added salt to the solution. According to our data, by adding salt to the water seemed to affect the cohesive properties of the water, as indicated by the severe reduction of the number of drops the penny would hold. We were not too surprised with our findings. We did notice some anomalies during our experiment, however. For instance, we did notice that we have a wide range of drops with the pure water solution, which ranged from 36-52. We think the lower number is more of the exception than the rule - as we noticed that the next number in series was closer to the higher number than the lower number. We think it might be because as we dropped the water, the drops may have been slightly bigger, or the drops touched the puddle on the penny, which reduced the number of drops. If we were to run this test again, we need to increase the height the drops that were administered to ensure that the drops don’t come in contact with the puddle while the water is still being formed from the pipet. The salt water solution, however, has a much tighter range of data - only having a four point difference and each of them in the lower to mid 20’s. What this means is that our hypothesis is thus far correct - in that the salt disrupts the cohesive properties of water. We still think that the reason the salt disrupts the cohesive property of water is due largely in part to the salt molecules slipping in between and separating the water molecules. We do know that water is polar and the salt becomes ionic when dissolved in water (Ophardt, 2003). Perhaps the adhesive attraction that water has with the salt is stronger than the cohesive property of water (Freeman, 2007). Further testing would be needed to suggest such a hypothesis by possibly using magnets or some other device with positive and negative charges to see how it affects the polarity of water & salt. |
Data Analysis:
Conclusion:
|
References
|
Perlman, H. (March 17, 2014) “Adhesion & Cohesion of Water” USGS.gov <http://water.usgs.gov/edu/adhsion.html> Last Retrieved August 18, 2014
Ophardt, C. (2003) “Solubility of Salts” Virtual Chembook, Elmhurst College <http://www.elmhurst.edu/~chm/vchembook/171solublesalts.html> Last Retrieved September 9, 2014 Freeman, S. (October 18, 2007) “How Water Works” HowStuffWorks.com <http://science.howstuffworks.com/environmental/earth/geophysics/h2o7.htm> Last Retrieved August 18, 2014 |
Author Last, F. (Date Published) "Title of the article written" Publisher Source <full web address> Last Retrieved Date
|